  
The atom is held together by electrostatic forces between the positive nucleus and negative surroundings. Also, the Bohrs theory couldnt explain the fine structure of hydrogen spectrum and splitting of spectral lines due to an external electric field (Stark effect). 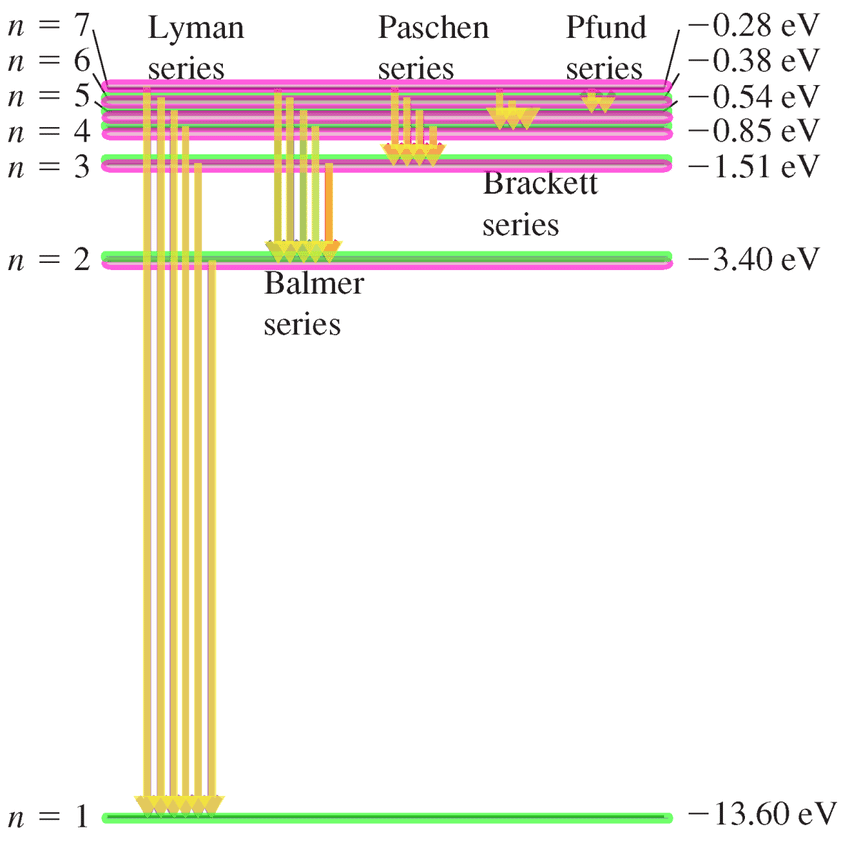
In the model, electrons orbit the nucleus in atomic shells. In 1913, Niels Bohr proposed the atomic Hydrogen hypothesis. The ground state energy atom and radius of Bohr hydrogen is derived under the condition that the basic theory has problems and the theoretical formula is set to, which has no experimental basis. He described it as a positively charged nucleus, comprised of protons and neutrons, surrounded by a negatively charged electron cloud. Niels Bohr introduced the atomic Hydrogen model in 1913. Bohr Model for Hydrogen Atom In an atom electron revolve around the nucleus in certain definite circular paths known as orbit or energy shells. The orbits are quantized (nonclassical) but are assumed to be simple circular paths (classical). In short, quantized energy levels release specific bands of light with unique colors. Bohrs model is what we call semiclassical. However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner.\) According to the Bohr model, the wavelength of the light emitted by a hydrogen atom when the electron falls from a high energy (n 4) orbit into a lower energy. Neils Bohr used the emission spectra of hydrogen to develop his model of the atom. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. The Bohr model of a hydrogen atom was proposed by scientist Neils Bohr in the year 1913. For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Thus, the Bohr model for heavier atoms described electron shells. Although the Bohr model is still used today, especially in elementary textbooks, a more sophisticated (and complex) model the quantum mechanical model is used much more frequently. Lesson Objectives Bohr Used Atomic Spectra to Develop His Model Electron Energy Is Quantized Bohr Used a Formula to Determine Allowed Energy Levels Atomic. the spectrum of an atom or ion containing one electron only. Once the level was full, additional electrons would be bumped up to the next level. The Bohr model works well for very simple atoms such as hydrogen (which has 1 electron) but not for more complex atoms. Bohr believed each electron orbit could only hold a set number of electrons. More electrons were required to cancel out the positive charge of all of these protons. o Electrons can also jump between these orbits either by absorbing or releasing photons. o He hypothesized that the electron was thus limited to certain orbits termed as discrete energy levels. Heavier atoms contain more protons in the nucleus than the hydrogen atom. o Bohr described the hydrogen atom in terms of an electron that moves in a circular orbit around a nucleus. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
